What Are Organoids?
Organoids are three-dimensional (3D) miniature structures cultured in vitro that closely mimic the architectural and functional features of their corresponding organs in vivo [1]. More specifically, organoids are formed through processes of self-renewal and self-organization, yielding structures that exhibit spatial organization similar to native organs and perform specialized physiological functions.
Organoids are characterized by three principal attributes. First, they demonstrate the ability of cells to self-organize via spatial arrangement and cellular differentiation, thereby replicating functional aspects of the original organ. Second, they consist of cell types representative of those found in the source organ [2]. Third, they are capable of performing certain organ-specific functions such as filtration, excretion, neural signaling, and contraction.
Historical Development of Organoids
The conceptual foundation of organoid technology traces back to early investigations into cellular self-organization. A notable example H.V. Wilson’s 1907 observation that dissociated sponge cells could reaggregate into functional multicellular organisms [3]. Subsequent advancements in vertebrate biology, coupled with the isolation of murine pluripotent stem cells (PSCs) in 1981, represented a seminal advance in stem cell biology [4, 5]. Further key developments included the derivation of human embryonic stem cells (ESCs) in 1998 [6] and the generation of induced pluripotent stem cells (iPSCs) in 2006 [7].
The contemporary era of organoid research commenced in 2009 with the generation of the first intestinal organoid from Lgr5+ stem cells by Hans Clevers [8]. This breakthrough catalyzed rapid expansion in the field, leading to the establishment of organoid models of the brain, kidney, lung, and other organs. These 3D systems now serve as indispensable tools in biomedical research, providing physiologically relevant alternatives to conventional two-dimensional (2D) cell cultures and animal models.
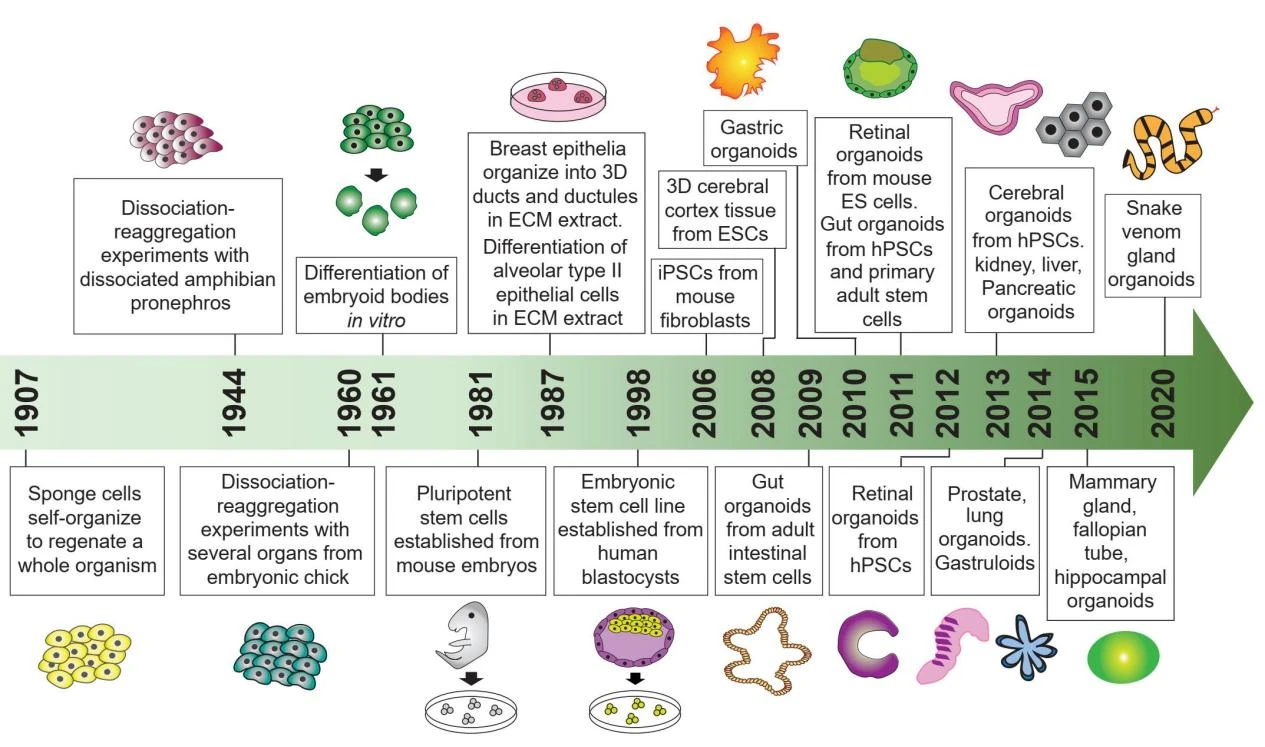
Figure 1. Timeline of key developments in organoid culture technologies, highlighting landmark studies and breakthroughs.
The Need for Alternative Model Systems in Biomedical Research
The ethical principles of the 3Rs—“Replacement, Reduction, and Refinement”—have guided animal research since their introduction in 1959. These principles mandate that animal experiments should only be conducted when no alternative methods are available. In recent years, regulatory agencies have increasingly acknowledged the utility of advanced in vitro models. For instance, in 2017, the U.S. Food and Drug Administration (FDA) incorporated data from organ-on-a-chip platforms into a new drug application for the first time. By August 2022, the FDA reviewed an investigational new drug application that included preclinical data derived from organ-on-a-chip technology, representing a pivotal advancement in alternatives to animal testing. More recently, the agency has moved to eliminate mandatory animal testing for monoclonal antibodies and certain other therapeutics. Consequently, the adoption of alternative model systems such as organoids has become increasingly imperative.
Generation and Preparation of Organoids
Organoids can be derived from either pluripotent stem cells (PSCs)—including embryonic stem cells (ESCs) and induced pluripotent stem cells (iPSCs)—or organ-specific adult stem cells (ASCs). These cells are cultured under precisely controlled conditions that promote recapitulation of innate developmental programs and self-organization into functional 3D structures. The presence of active stem cell populations in organoid cultures enables substantial expansion and long-term maintenance.
A typical organoid culture system requires several critical components: a suitable extracellular matrix (ECM), signaling factors essential for maintaining the stem cell niche, and differentiation-inducing agents. The ECM, often composed of proteins such as laminin, collagen, nidogen, and fibronectin, provides structural support and biochemical cues necessary for 3D organization. Niche maintenance factors primarily facilitate cell proliferation and suppress apoptosis. Moreover, patient-derived tumor organoids offer a clinically relevant platform for personalized medicine approaches.
Categories and Applications of Organoids
Since the successful generation of intestinal organoids in 2009, organoid technology has been extended to a wide array of organs. The significance of this methodology was underscored when organoids were listed among the “Top 10 Technologies” by Science in 2013 and named “Method of the Year” in 2017 by Nature Methods. To date, organoids have been established for numerous organs, including the brain, inner ear, optic cup, lung, intestine, kidney, liver, pancreas, stomach, and others. These systems model both normal and pathological tissues, including cancer.
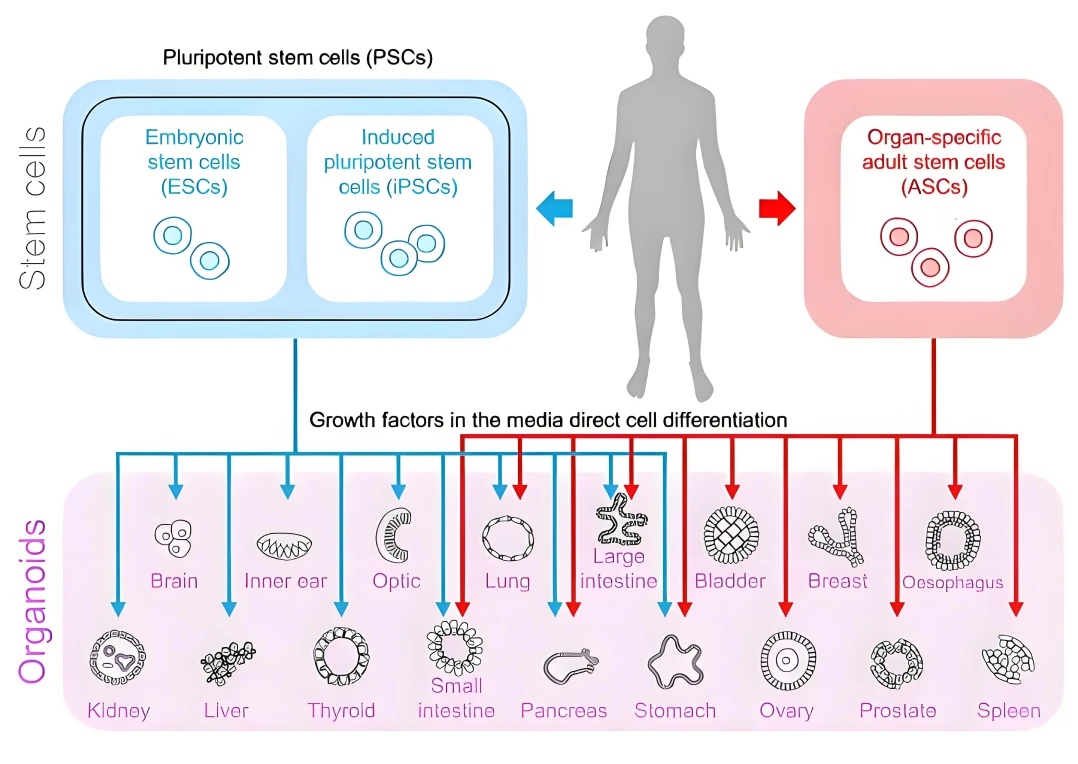
Figure 2. Schematic representation of organoid establishment from human or animal pluripotent stem cells (PSCs) and adult stem cells (ASCs).
Although organoids derived from human PSCs or ASCs are not exact replicas of human organs, they faithfully recapitulate key structural and functional attributes of in vivo tissues while permitting long-term culture and expansion. As such, they provide near-physiological model systems for applications in genomics, infectious disease, developmental biology, and disease modeling. Furthermore, patient-derived organoids (PDOs) offer promising platforms for drug screening, biomarker identification, and regenerative medicine.
Comparison of Organoids with Conventional Model Systems
Traditional models for studying disease mechanisms and drug development include 2D cell lines and animal models—primarily murine and non-human primates (NHPs). While widely used, these systems exhibit significant limitations. 2D cultures, though facile to manipulate, often consist of homogeneous cell populations that lack the heterogeneity and genomic stability of original tissues. Mouse models, though prevalent, suffer from inherent physiological differences from humans, particularly in areas such as drug metabolism, immunology, and neurobiology. NHPs, while phylogenetically similar to humans, entail high costs, ethical concerns, and operational challenges that preclude high-throughput applications.
In contrast, human stem cell-derived organoids offer several advantages: they are genetically stable, amenable to genome editing, suitable for high-throughput screening, and can be biobanked for future use. They also exhibit high clinical relevance and can be rapidly generated—often within one to two weeks—enabling expedited drug screening pipelines [9].
Notably, PDOs retain phenotypic and genotypic characteristics of original patient tumors. Their predictive validity was highlighted in a landmark study by Vlachogiannis et al., which demonstrated that PDO responses to therapeutics correlated strongly with clinical outcomes in patients with metastatic gastrointestinal cancers [10]. These findings underscore the potential of PDOs in personalized medicine and drug development.
In summary, organoid technology narrows the gap between conventional models and human physiology while providing a stable, scalable, and ethically tenable platform for investigating genetic diversity, disease mechanisms, and drug responses.
Challenges in Organoid Technology
Despite their promise, organoids face several limitations. A primary constraint is the general absence of vascularization, which restricts nutrient delivery and waste removal, ultimately limiting organoid size and viability. To address this, studies have explored co-culture systems incorporating endothelial cells to promote vascular network formation.
Another challenge is the lack of immune components in most current organoid models. Co-culture protocols integrating immune cells have been developed to better mimic tumor-immune interactions and enhance physiological relevance [11], such as tumor organoid co-cultures with T cells or macrophages. For instance, immune cells may be introduced into organoid cultures via direct co-culture or through the addition of recombinant cytokines.
Furthermore, most existing organoid models represent isolated organs and cannot capture systemic drug effects. To overcome this, multi-organoid systems connected via microfluidic perfusion circuits have been developed to more accurately model drug toxicity and efficacy across different organs [12].
Together, advances in vascularization, immune integration, and systemic modeling are actively addressing these limitations, which will further enhance the physiological accuracy and translational potential of organoid models.
Conclusion
Although organoid technology is still evolving, it holds significant potential across numerous fields including developmental biology, disease modeling, drug discovery, and regenerative medicine. As a human-derived, scalable, and ethically advantageous platform, organoids are poised to reduce reliance on animal testing and open new avenues for understanding human biology and disease.
Instead of animal models, these miniature organ-like structures are expanding their applications in basic, translational, and clinical research, promising to accelerate scientific discovery and therapeutic development.
References
[1] H. Clevers, Modeling Development and Disease with Organoids, Cell 165(7) (2016) 1586-1597.
[2] K.R. Stevens, K.L. Kreutziger, S.K. Dupras, F.S. Korte, M. Regnier, V. Muskheli, M.B. Nourse, K. Bendixen, H. Reinecke, C.E. Murry, Physiological function and transplantation of scaffold-free and vascularized human cardiac muscle tissue, Proceedings of the National Academy of Sciences of the United States of America 106(39) (2009) 16568-16573.
[3] H.V. Wilson, A new method by which sponeges may be artificially reared, Science (New York, N.Y.) 25(649) (1907) 912-915.
[4] G.R. Martin, Isolation of a pluripotent cell line from early mouse embryos cultured in medium conditioned by teratocarcinoma stem cells, Proceedings of the National Academy of Sciences of the United States of America 78(12) (1981) 7634-7638.
[5] M.J. Evans, M.H. Kaufman, Establishment in culture of pluripotential cells from mouse embryos, Nature 292(5819) (1981) 154-156.
[6] J.A. Thomson, J. Itskovitz-Eldor, S.S. Shapiro, M.A. Waknitz, J.J. Swiergiel, V.S. Marshall, J.M. Jones, Embryonic stem cell lines derived from human blastocysts, Science (New York, N.Y.) 282(5391) (1998) 1145-1147.
[7] K. Takahashi, S. Yamanaka, Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors, Cell 126(4) (2006) 663-676.
[8] T. Sato, R.G. Vries, H.J. Snippert, M. van de Wetering, N. Barker, D.E. Stange, J.H. van Es, A. Abo, P. Kujala, P.J. Peters, H. Clevers, Single Lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche, Nature 459(7244) (2009) 262-265.
[9] M. Li, J.C. Izpisua Belmonte, Organoids - Preclinical Models of Human Disease, The New England journal of medicine 380(6) (2019) 569-579.
[10] G. Vlachogiannis, S. Hedayat, A. Vatsiou, Y. Jamin, J. Fernández-Mateos, K. Khan, A. Lampis, K. Eason, I. Huntingford, R. Burke, M. Rata, D.M. Koh, N. Tunariu, D. Collins, S. Hulkki-Wilson, C. Ragulan, I. Spiteri, S.Y. Moorcraft, I. Chau, S. Rao, D. Watkins, N. Fotiadis, M. Bali, M. Darvish-Damavandi, H. Lote, Z. Eltahir, E.C. Smyth, R. Begum, P.A. Clarke, J.C. Hahne, M. Dowsett, J. de Bono, P. Workman, A. Sadanandam, M. Fassan, O.J. Sansom, S. Eccles, N. Starling, C. Braconi, A. Sottoriva, S.P. Robinson, D. Cunningham, N. Valeri, Patient-derived organoids model treatment response of metastatic gastrointestinal cancers, Science (New York, N.Y.) 359(6378) (2018) 920-926.
[11] C.M. Cattaneo, K.K. Dijkstra, L.F. Fanchi, S. Kelderman, S. Kaing, N. van Rooij, S. van den Brink, T.N. Schumacher, E.E. Voest, Tumor organoid-T-cell coculture systems, Nature protocols 15(1) (2020) 15-39.
[12] A. Skardal, S.V. Murphy, M. Devarasetty, I. Mead, H.W. Kang, Y.J. Seol, Y. Shrike Zhang, S.R. Shin, L. Zhao, J. Aleman, A.R. Hall, T.D. Shupe, A. Kleensang, M.R. Dokmeci, S. Jin Lee, J.D. Jackson, J.J. Yoo, T. Hartung, A. Khademhosseini, S. Soker, C.E. Bishop, A. Atala, Multi-tissue interactions in an integrated three-tissue organ-on-a-chip platform, Scientific reports 7(1) (2017) 8837.

